Improving Patient Centricity through effective usage of Real-World Evidence Derived from Real-World Data.
Patient centricity in healthcare is prioritized globally, utilizing Real-world Data (RWD) and Real-World Evidence (RWE). RWD is collected from everyday practices, while RWE offers insights for decision-making, improving outcomes and regulatory processes. Stakeholders leverage RWE for treatment plans, coverage decisions, clinical trials, and personalized care. Challenges include data quality and privacy concerns. Addressing these challenges enables effective use of RWE, enhancing patient-centric healthcare decision-making.

Patient centricity has become a global priority in healthcare, with a growing focus on utilizing Real-world data (RWD) and Real-World Evidence (RWE) to inform decision-making. RWD refers to data collected from everyday clinical practices and non-clinical sources, such as electronic medical records, health checkups, patient-generated data, insurance claims, and wearable devices. RWE is the scientific evidence obtained by analyzing RWD, and it offers several potential benefits in enhancing patient outcomes, regulatory approvals, reimbursement, and health technology assessment decision-making.
Across the healthcare ecosystem, RWE has been utilized by various stakeholders to support decision-making. Regulators, such as the FDA and EMA, use RWD and RWE to monitor safety, make regulatory decisions, and assess post-market outcomes. Healthcare providers use RWE to inform treatment plans, clinical practice guidelines, and clinical decision support tools. Payers rely on RWE for coverage decisions and reimbursement of claims. Health technology assessment bodies use RWE to make reimbursement recommendations, and pharmaceutical companies use RWE for drug discovery, clinical trial design, and commercialization strategies.
One significant way RWE can improve patient centricity is by streamlining the clinical trial recruitment and participation process. RWD and RWE can support regulatory filings, identify eligible patients through EHR systems, and alert physicians about suitable patients for clinical trials. This can accelerate the clinical trial process, expand patient access, and enhance patient engagement in research. Another benefit of RWE in patient centricity is its ability to provide clinically-rich insights into everyday practice. RWE can help stakeholders understand what happens in real-world settings, why specific treatments are effective, and how interventions can be tailored to individual patients. This can lead to more personalized, patient-centered care, improved outcomes, and satisfaction.
Stakeholders Use RWD/RWE for Decision-Making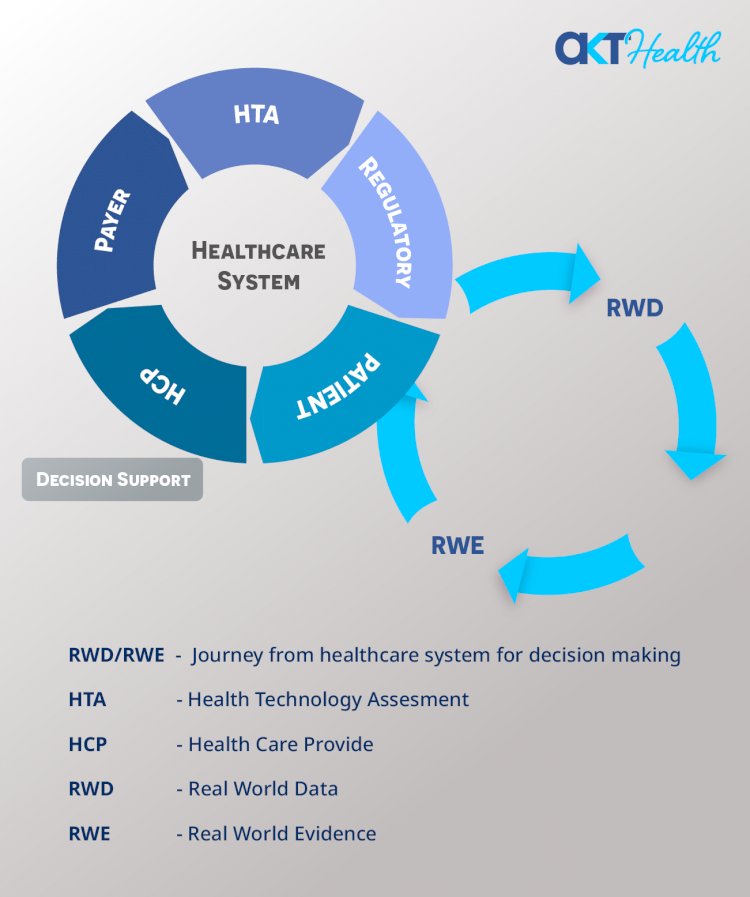
RWE can contribute to evidence-based decision-making in healthcare. By leveraging RWD and RWE, stakeholders can make informed decisions on treatment plans, coverage, reimbursement, and regulatory approvals. This can lead to more efficient and effective healthcare delivery, with a focus on patient outcomes and preferences.
- Healthcare providers use RED/RWE for decisions on treatment plans and orders of tests, procedures, and prescriptions for individual patients and also to adopt clinical practice guidelines and develop clinical decision support tools.
- Payers use RWD/ RWE for decision-making on drug formularies, Health Plan coverage, and Reimbursements of individual claims.
- Regulatory agencies such as FDA could review RWD/RWE as a supplement to RCT evidence of Marketing authorization, to monitor post-market Safety and adverse events.
- Health Technology Assessment (HTA) bodies could review RWD/RWE to make positive or negative reimbursement recommendations to payers.
- Pharmaceutical companies could use these data in their internal decision-making for:
- Drug discovery and preclinical development
- Clinical trial design and Interpretation
- Innovative, new treatment in the real world
- Commercialization
However, there are challenges in utilizing RWE effectively, such as data quality, data interoperability, privacy concerns, and potential biases in real-world data. A rigorous methodology, standardization, and validation are crucial in ensuring the reliability and validity of RWE. The effective usage of RWE derived from RWD can contribute to improving patient centricity in healthcare. By leveraging RWE, stakeholders can make informed decisions, streamline clinical trials, and provide personalized and patient-centered care. However, addressing challenges and ensuring data quality and validity are essential for the successful utilization of RWE in healthcare decision-making.